Research
Overview
Development of functional molecules and advanced synthetic methods to synthesize them
As organic chemists, "manipulating molecules to confer functionality" is a privilege granted to us.
The number of molecules is literally infinite. There are countless molecules that have yet to be discovered or synthesized, hidden in this treasure trove. Our research focuses on discovering molecules that are useful for society or intriguing, even if they do not have direct practical applications.
We have been interested in light-related chemical reactions and molecules. For example, we have been developing photochemical reactions that absorb sunlight to convert carbon dioxide into other molecules (artificial photosynthesis), molecules that emit light in various colors, and molecules that change their luminescent color or brightness in response to stimuli. Light is the only energy source on Earth that can be used semi-permanently. We aim to effectively utilize its benefits and promote environmentally benign chemistry.
Development of photochemical organic reactions and light-responsive molecular systems
Photochemical Reaction
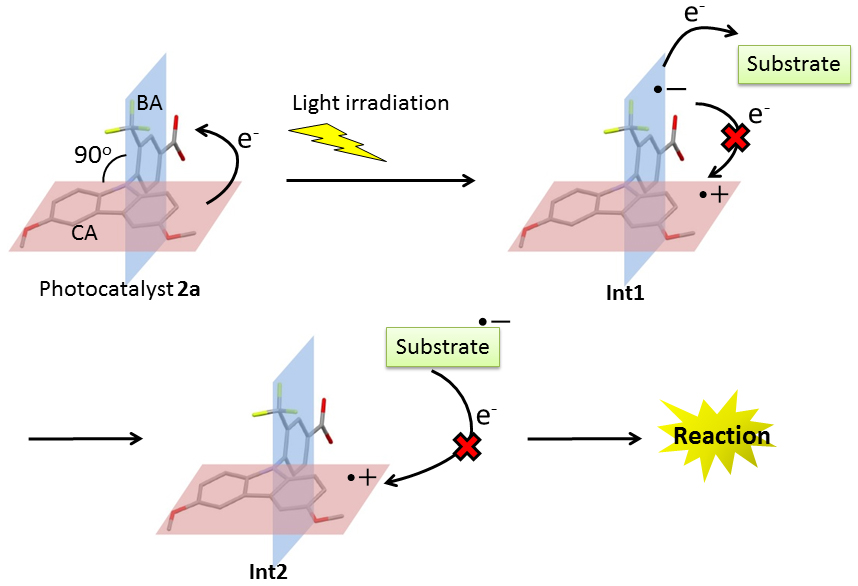
Design of Unprecedented Photocatalyst
Light is so strong energy source that it can cleave chemical bonds in a molecule. However, it does not follow that light can cleave any bond in a molecule, because a certain specific structure is required to absorb and utilize energy from the light.
We are currently developing organic photocatalyst which can absorb light and enable a chemical reaction that could not be realized thermally.
Synthesis of luminescent materials

Development of charge-separated photocatalysts
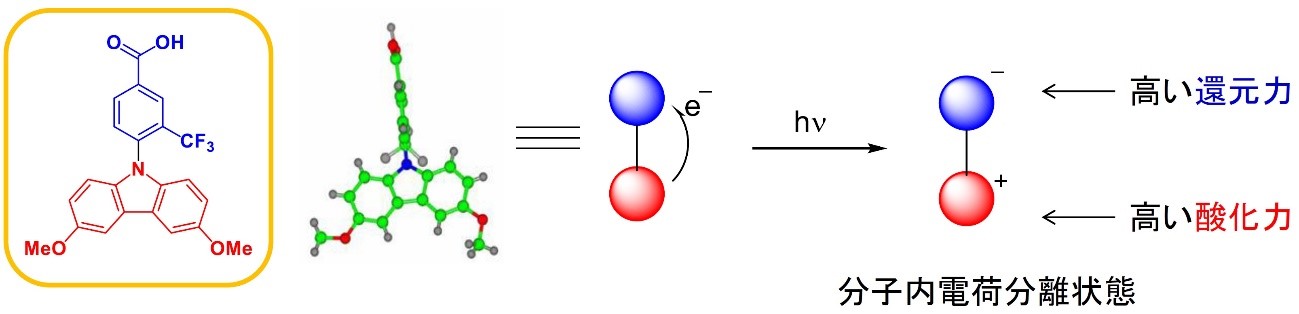
(This quote is cited from Atlas of Science)
https://atlasofscience.org/perpendicular-plane-configuration-to-prevent-backward-electron-transfer/
Photoirradiation systems used in our laboratory




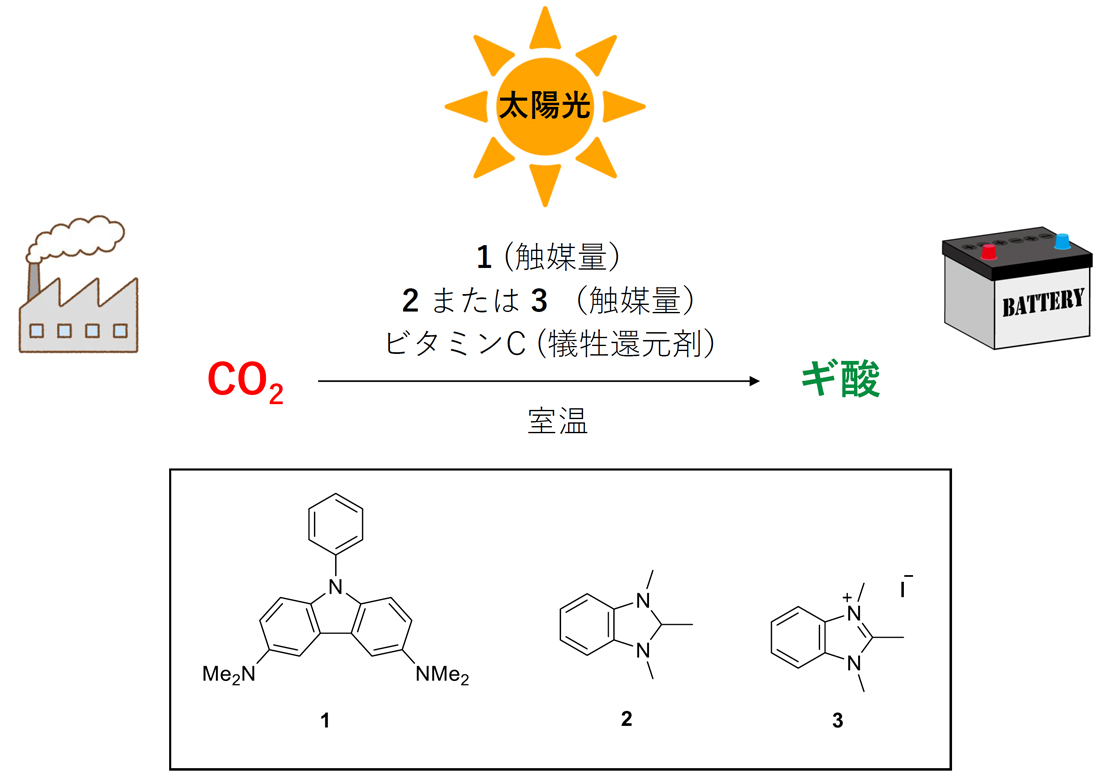
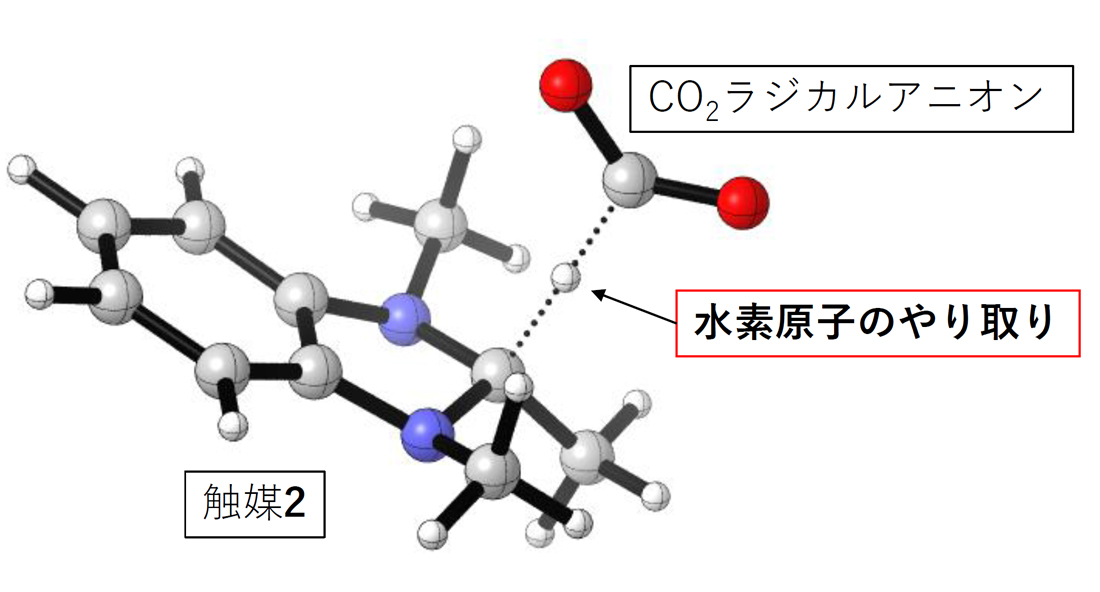
Artificial Photosynthesis without Transition Metal
Development of Artificial Photosynthesis without using Transition Metals
In natural photosynthesis, solar energy is used to synthesize oxygen and glucose from water and carbon dioxide. Thanks to photosynthesis, animals including humans are able to respire and move by obtaining nutrients.
Artificial photosynthesis, which mimics natural photosynthesis, is a technology that uses light energy to artificially produce hydrogen and oxygen from water, or convert carbon dioxide into methane. With the depletion of fossil fuels becoming a pressing issue, artificial photosynthesis is an essential technology for achieving a sustainable society. Artificial photosynthesis has been extensively studied, and there are many reported examples. However, in previous examples, transition metal elements have almost always been included in catalyst molecules used in the reaction process of artificial photosynthesis. This is because transition metal elements are good at catalyzing the oxidation and reduction steps involved in artificial photosynthesis reactions. However, transition metal elements are often rare and subject to country risks, posing challenges from the perspective of sustainability.
In recent years, our laboratory has developed one approach of artificial photosynthesis that does not use any transition metal elements, by assigning the roles that transition metals typically play to non-transition metal molecules. We continue to aim for the development of new reaction systems that challenge the inherent properties of elements and rely on precise design of organic molecule structures to achieve highly challenging reactions, testing the pride of being organic chemists.
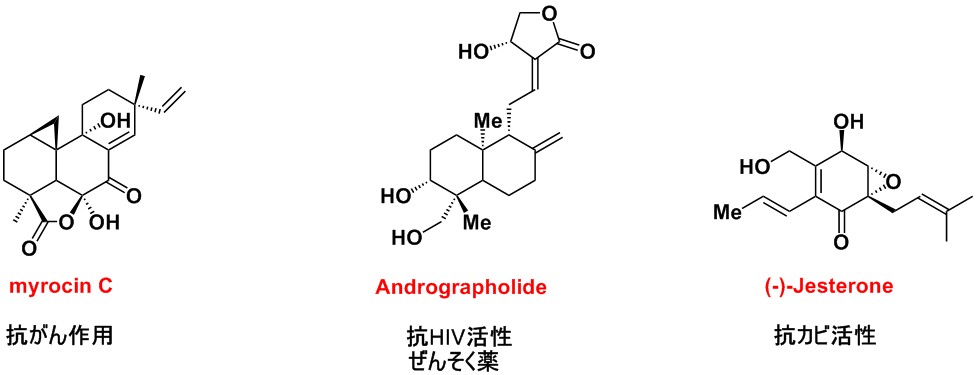
Synthesis of bioactive molecules and development of synthetic methodologies to access them
Drug Discovery
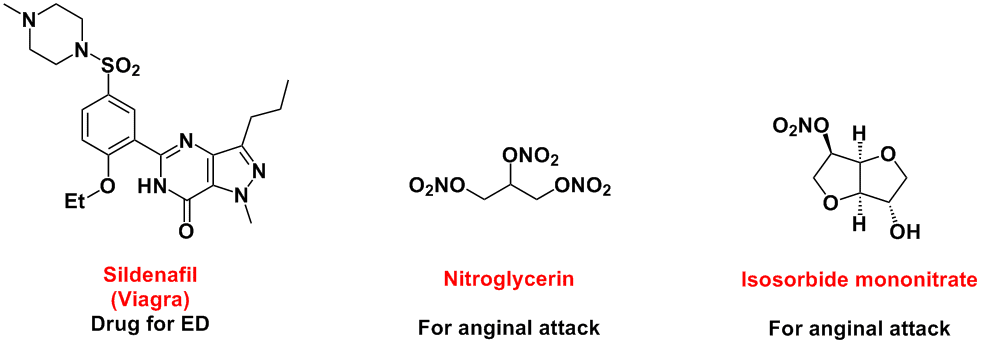
Development of Nitric Oxide Donor -Providing nitric oxide at right time at right place with right amount-
Nitric oxide (NO) has various important biological functions, including activation of the soluble guanylate cyclase, which leads to vascular dilation and the inhibition of platelet aggregation. Examples of medicines which target a NO molecule are nitroglycerin and sildenafil (Viagra).
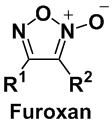
NO is also known to function as a neurotransmitter to be involved in learning and memory process, so attracts attention as a target for the treatment of epilepsia and Alzheimer disease. In our lab, we are developing NO donors with novel scaffolds. Our research goal is the discovery of the NO donors which release NO in control of time, amount and place. One of the NO donor molecules we are now focused on is furoxan, which has a unique molecular structure as shown below.
Total Synthesis
Total Synthesis — Creating Molecules Learned from Nature
Total synthesis is a field of research in which a target molecule—typically one with a highly complex structure—is painstakingly constructed step by step from readily available simple compounds, such as those found in reagent catalogs. The targets often include bioactive compounds discovered in nature, such as pharmacologically active natural products or trace physiologically active substances produced by living organisms.
In total synthesis, chemists draw on their full arsenal of organic chemistry knowledge, experience, and intuition to “nurture” molecules toward the final goal. Along the way, they frequently encounter troublesome “wild” intermediates with challenging properties: instability toward light or air, a tendency to react with themselves, or poor solubility in common solvents. Unexpected side reactions—those very much not desired—also occur with frustrating regularity. By overcoming these difficulties one by one, chemists ultimately advance toward the synthesis of the final target molecule.
Recently, we accomplished the total synthesis of several ant pheromones and, through collaboration with a biological research group, gained new insights into ant social behavior and ecology.